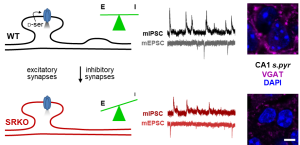
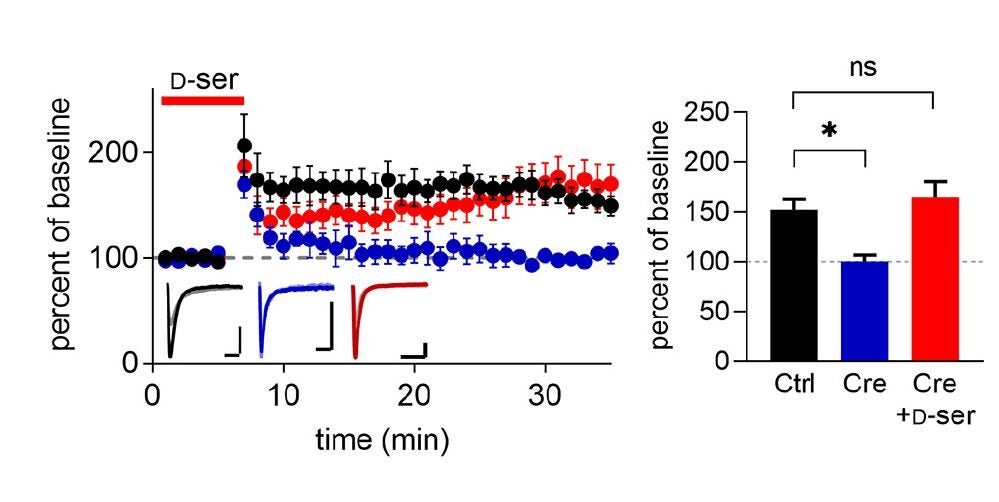
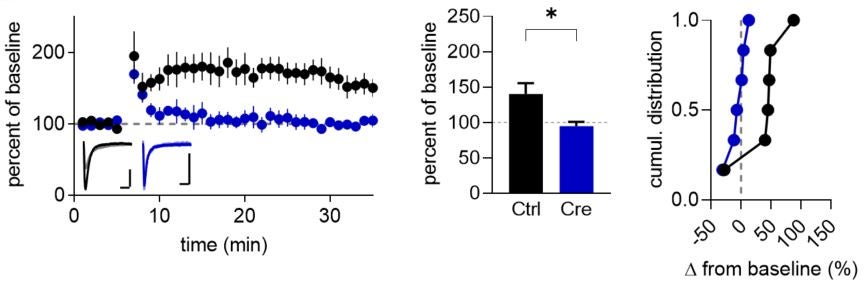
Congrats to Eden for her finally published dissertation work!
NMDARs have been found to drive synaptic plasticity via non-ionotropic (ion flux-independent) signaling upon the binding of glutamate in the absence of co-agonist, though conflicting results have led to controversy. Here, we found that D-serine inhibits non-ionotropic NMDAR-mediated long-term depression (LTD) and LTD-associated spine shrinkage. Thus, the developmental regulation of D-serine is likely a determinant of non-ionotropic NMDAR plasticity during critical periods of plasticity. In addition, these findings suggest that a major source of the contradictory findings among labs might be attributed to experimental variability in D-serine availability. Read the paper here.