I was recently interviewed for a podcast by Nino Ramirez, the editor-in-chief for the Journal of Neurophysiology, about our paper “Increased excitation-inhibition balance & loss of GABAergic synapses in the serine racemase knockout model of NMDA receptor hypofunction.” It was
PP1 dephosphorylates GluN2B in the PDZ ligand to regulate synaptic NMDA receptor content
In this ongoing collaboration with Antonio Sanz-Clemente at Northwestern University, we define an additional mechanism regulating the synaptic expression of the GluN2B subunit of NMDA receptors. Following synaptic activity, the GluN2B subunit is phosphorylated on the C-terminal PDZ ligand at
Long-Term Depression Is Independent of GluN2 Subunit Composition

Excited to see the lab’s first original research publication in print and congrats to Jon Wong for his first scientific publication! It was even a featured article. PDF can be found here.
New review article on non-ionotropic NMDA receptor signaling
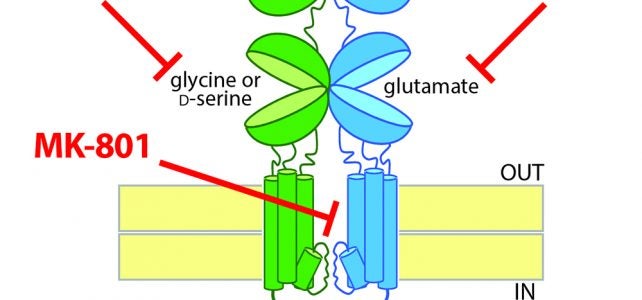
Gray JA, Zito K, and Hell JW (2016). Non-ionotropic signaling by the NMDA receptor: controversy and opportunity. F1000Research, 5(F1000 Faculty Rev): 1010. In collaboration with Karen Zito and Johannes Hell, I recently reviewed the emerging literature on possible non-ionotropic signaling
My first paper at UC Davis
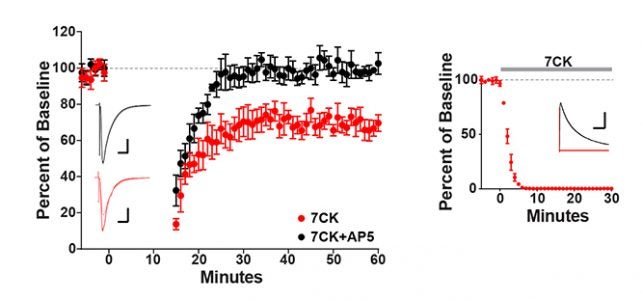
Was excited to be able to contribute to an elegant study from the Zito Lab showing that NMDA receptor-mediated spine shrinkage requires glutamate binding but not ion flux through the channel, supporting recent studies showing that the NMDA receptor can

