We all went to a reception at the Willis Tower in Chicago during the 2019 SfN meeting.
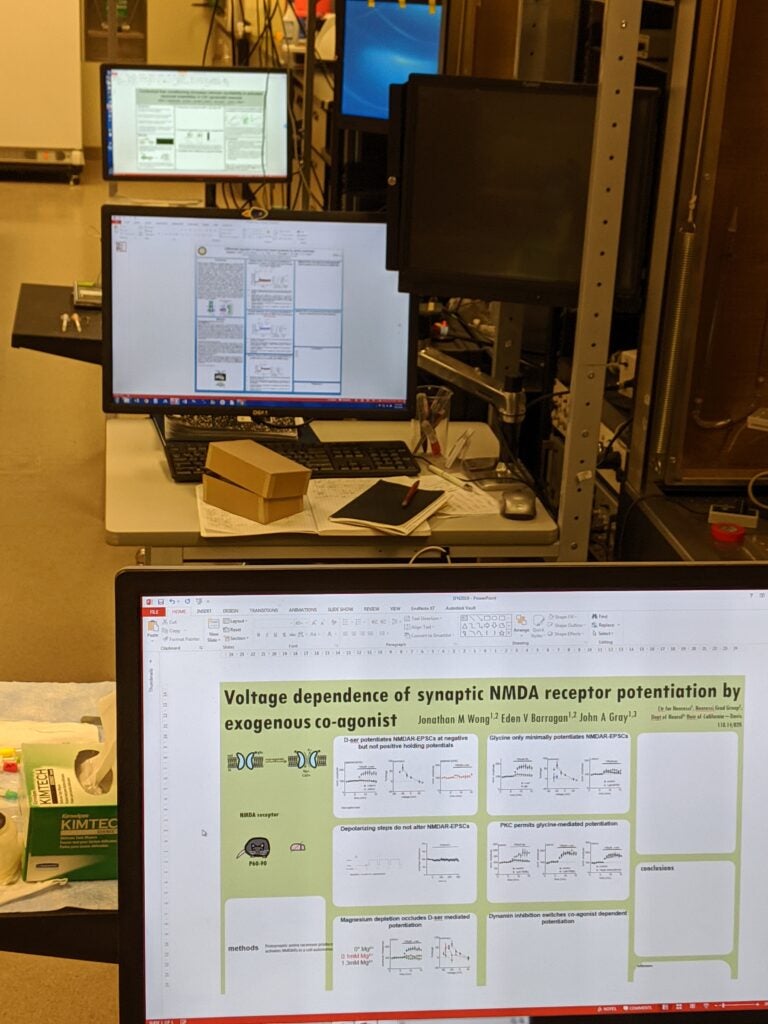
SfN 2019 Poster Prep
PP1 dephosphorylates GluN2B in the PDZ ligand to regulate synaptic NMDA receptor content
In this ongoing collaboration with Antonio Sanz-Clemente at Northwestern University, we define an additional mechanism regulating the synaptic expression of the GluN2B subunit of NMDA receptors. Following synaptic activity, the GluN2B subunit is phosphorylated on the C-terminal PDZ ligand at S1480 but casein kinase 2 (CK2) resulting in the removal of GluN2B from the synapse. We now show that these extrasynaptic GluN2B subunits are dephosphorylated by PP1 in an activity-dependent manner resulting in the re-entry into the synapse.
See our paper at Cell Reports: https://doi.org/10.1016/j.celrep.2019.06.030

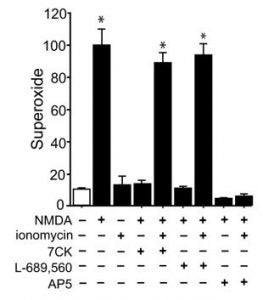
Parallel non-ionotropic and ionotropic requirements for NMDA-mediated superoxide production
In this collaboration with the Swanson lab at UCSF, we show that NMDA-mediated superoxide production requires both a source of calcium influx and agonist binding to the glutamate site of the NMDA receptor (GluN2B subunit to be specific). Thus, both Ca2+ and agonist -binding are necessary, but neither is sufficient. This is the first report demonstrating this dual requirement and I predict this will be rule rather than the exception for NMDA receptor function.
https://www.nature.com/articles/s41598-018-35725-5
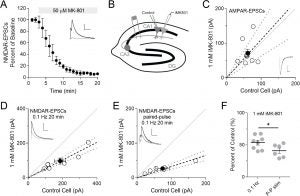
Intracellular MK801 does not fully block NMDA receptors
MK801 is an open-channel blocker of the NMDA receptor pore. That is, when the receptor is activated and the channel opens, MK801 enters the pore and blocks it. Usually, MK801 is used extracellularly, but multiple studies have included it in the intracellular pipette solution during whole-cell patch clamp recordings. We show here that MK801 has a much lower effective affinity for NMDA receptors when used intracellularly, suggesting caution is needed when interpreting such studies.
Congrats to Eden passing her qualifying exam!
Psychedelics Promote Structural and Functional Neural Plasticity
 Exciting new collaborative publication with David Olson’s lab on the possibility that psychedelics may be rapid-acting antidepressants. Was fun to get back into studying 5-HT2A receptors after a long hiatus.
Exciting new collaborative publication with David Olson’s lab on the possibility that psychedelics may be rapid-acting antidepressants. Was fun to get back into studying 5-HT2A receptors after a long hiatus.
Covered by the press:
UC Davis
LA Times
Newsweek
New Scientist
Wall Street Journal
Metaplasticity contributes to memory formation in the hippocampus
 After initial learning, subsequent learning is independent of NMDA receptor function, but requires mGluRs and increases in intrinsic excitability. New publication in collaboration with the Wiltgen Lab.
After initial learning, subsequent learning is independent of NMDA receptor function, but requires mGluRs and increases in intrinsic excitability. New publication in collaboration with the Wiltgen Lab.
https://www.ucdavis.edu/news/how-experience-changes-basics-memory-formation/
Long-Term Depression Is Independent of GluN2 Subunit Composition
Excited to see the lab’s first original research publication in print and congrats to Jon Wong for his first scientific publication! It was even a featured article. PDF can be found here.